Amyloid-? (1-8, A2V) Peptide
Amyloid-? (1-8, A2V) Peptide
Amyloid-? (1-8, A2V) is a truncated form of amyloid-? (A?) that contains a valine to alanine substitution at position 2...
€525.00
Enzyme ImmunoAssay (EIA) is a technique to detect and quantify antigens (proteins, hormones…) or antibodies in samples. It relies on the ability of an antibody to bind a specific antigen. Either the antibody or the antigen is labelled with an enzyme whose substrate is a chromogen or a fluorogen converted in a measurable product (color or fluorescence).|Enzyme-linked Immunosorbent Assay (ELISA) is a type of EIA using a solid phase (ex: microtiter plate) coated with an antigen immobilizing the molecule to detect. Over the time, scientists have extended the term ELISA to EIAs using an antibody coating the solid phase. That explains why our EIA kits using coated antibodies are also called ELISA kits.|Bertin Bioreagent’s expertise is to develop analytical tools for biomarkers. As such, in 2004, Bertin Bioreagent launched its first Ghrelin Biomarker assay kits, under the SPI-Bio brand name, as a result of its state-of-the-art R&D research teams. Bertin Bioreagent was the first company providing bioanalytical tools to assay Acylated and Non-acylated Ghrelin with very high sensitivity.|Ghrelin discovered in 1999, is fast becoming an endocrinology target of the millennium. Ghrelin, identified in rat stomach as an endogenous ligand for the GH secretagogue receptor, is mainly produced in stomach, but has been demonstrated in many other organs. In addition to GH-releasing properties and its orexant action, ghrelin could act as an hormone having effects on gastric motility (similarity with the peptide hormone motilin), acidic secretion, cardiovascular action, antiproliferative effects, pancreatic and glucose metabolism function, sleep… Ghrelin gene raises to mRNA prepro-ghrelin of 117 amino acids. This precursor is processed into ghrelin, 28 amino acids (human). Before being secreted, this peptide is octanoylated at Ser 3. This step is essential for biological activity. If the endogenous peptide appears directly related to feeding behaviour, the potential therapeutic importance of this hormone is not restricted to a regulator of food intake but also may be involved osteoporosis somatopaus, infertility and ovulation induction, and some cardiovascular diseases.
Territorial Availability: Available worldwide directly through Bertin or your local distributor
Technical Warning: Check the Additional Items Required section of this kit booklet to verify if UltraPure Water (Milli-Q or equivalent) is needed for this assay
| Size | 96 wells |
|---|---|
| Shipping | wet ice |
| Specificity | Human & rat ghrelin (17-28): <0.001%, Human acylated Ghrelin EIA: 118%, Human ghrelin (1-14): <0.001%, Human, mouse & rat ghrelin (Des-Octanoyl-Ser3): <0.001%, Rat acylated ghrelin: 100%, Rat ghrelin (1-11): <0.001% |
| Application media | Blood collection on inhibitor then 1/5 dilution prior assay, Plasma, buffer |
| Sample volume | 20 µL |
| Tracer | AcetylCholinesterase AChE |
| Detection limit | |
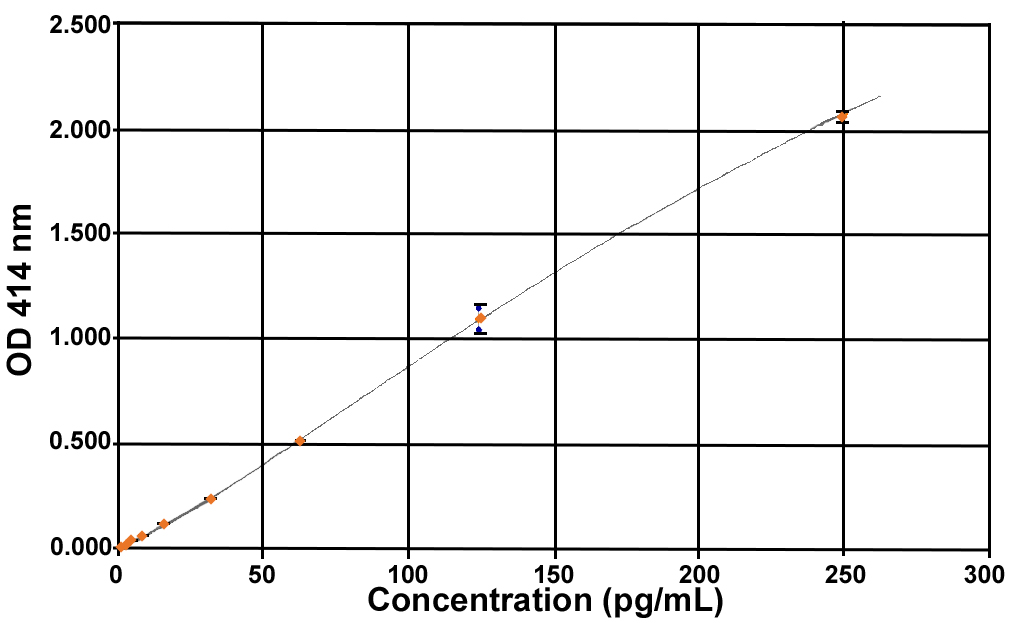
| Standard Curve Range | 2-250 pg/mL |
| Custom code | 3822000000 |
| UNSPSC code | 41116104 |
Amyloid-? (1-8, A2V) is a truncated form of amyloid-? (A?) that contains a valine to alanine substitution at position 2...
The cyclopentenone prostaglandin HPLC mixture contains all of the major UV-absorbing cyclopentenone prostaglandins and their precursors supplied in methyl acetate....
Programmed cell death protein 4 (PDCD4) levels are elevated during apoptosis and absent in many cancer samples.{16340,16167} Loss of PDCD4...
Histone H2A is a core histone that forms a dimer with histone H2B.{46631} Two histone H2A/H2B dimers form an octameric...
To be used in conjunction with Cayman’s FABP3 polyclonal antibody (Catalog No. 10233) to block protein-antibody complex formation during immunochemical...
This mixture contains the characteristic metabolites of both PGI2 and TXA2. Contents: Thromboxane B2, 11-dehydro Thromboxane B2, 6-keto Prostaglandin F1?,...
This mixture contains the primary COX products produced by most mammalian tissues. Contents: Prostaglandin D2, Prostaglandin E2, 6-keto Prostaglandin F1?,...
Platelet-activating factor (PAF) is an important lipid mediator involved in inflammation. PAF-acetylhydrolase (PAF-AH) is an extracellular phospholipase A2 which hydrolyzes...
Sirtuin 5 (SIRT5) is an enzyme that catalyzes the NAD-dependent removal of malonyl, succinyl, and glutaryl groups from target proteins.{55107,55108}...
Proprotein convertase subtilisin kexin 9 (PCSK9) is a member of the subtilisin serine protease family with an important role in...
The Cayman COX Inhibitor Pack contains a combination of frequently used cyclooxygenase (COX) inhibitors. Each kit contains aspirin, the archetype...
GST-tag Polyclonal Antibody (FITC) is a probe for the immunochemical detection of GST tags on recombinant proteins. Recombinant proteins are...
This mixture contains primary prostaglandins produced from arachidonic acid and dihomo-?-linolenic acid. Contents: Prostaglandin E1, Prostaglandin E2, Prostaglandin F1?, 6-keto...
TAF10 is one of many protein factors or coactivators associated with RNA polymerase II activity.{16903} One vial of this peptide...
This mixture contains the primary metabolites of prostaglandins (PGs) D2, E2, and F2?. Contents: 13,14-dihydro-15-keto PGD2, 13,14-dihydro-15-keto PGE2, 11?-PGF2?, 13,14-dihydro-15-keto...
Prostaglandin I synthase (PGIS) catalyzes the isomerization of PGH2 to PGI2. PGI2 (prostacyclin) is a potent vasodilator and inhibitor of...
To be used in conjunction with Cayman’s NAPE-PLD polyclonal antibody (aa 6-20) (Item No. 10306) to block protein-antibody complex formation...
Inositol hexakisphosphate kinase 2 (IP6K2) is a cytoplasmic kinase that catalyzes the conversion of IP6 to diphosphoinositol pentakisphosphate in the...