Territorial Availability: Available through Bertin Technologies only in France
- Synonyms
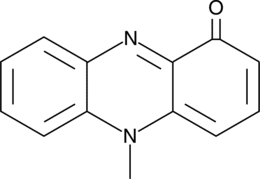
- 5-methyl-1(5H)-phenazinone
- Correlated keywords
- biochemicals cytokines free radical generators apoptosis inflammation oxidative injury infectious disease gene regulation immunology receptor agonists cystic fibrosis patients infected P aeruginosa pseudomonas aryl hydrocarbon receptor activates expression detoxifying enxymes cytokines clearance clears bacteria accelerate neutrophil in vitro acute inflammation bacteria bacterial survival in vivo lungs immunocompromised premature cellular senescence mammal nonenzymatic reduction NADPH hydrogen peroxide intracellular glutathione stress cells
- Product Overview:
Pseudomonas aeruginosa (P. aeruginosa) is a common pathogen affecting immunocompromised patients with acute diseases such as pneumonia and vasculitis or chronic diseases such as cystic fibrosis. It produces several phenazine toxic metabolites, the most predominant of which is a blue pigment, pyocyanin. Pyocyanin, which can reach concentrations of 100 µM in cystic fibrosis patients infected with P. aeruginosa, activates the aryl hydrocarbon receptor with a Ki value of 5.4 µM.{27028} This induces the expression of both detoxifying enzymes, resulting in pyocyanin degradation, and cytokines that facilitate the clearance of bacteria.{27028} Pyocyanin has been shown to accelerate neutrophil apoptosis in vitro, resulting in resolution of acute inflammation, which is beneficial for bacterial survival. It also induces a 10-fold acceleration of neutrophil apoptosis in vivo.{14843} Pyocyanin production results in reduced bacterial clearance from the lungs of immunocompromised patients. It has also been reported to induce apoptosis in human lung epithelial cells and to induce premature cellular senescence in mammalian cells.{14999} Pyocyanin undergoes nonenzymatic reduction by NADPH, which produces hydrogen peroxide and depletes intracellular glutathione levels, causing oxidative stress in susceptible cells.{14844}
Cayman Chemical’s mission is to help make research possible by supplying scientists worldwide with the basic research tools necessary for advancing human and animal health. Our utmost commitment to healthcare researchers is to offer the highest quality products with an affordable pricing policy.
Our scientists are experts in the synthesis, purification, and characterization of biochemicals ranging from small drug-like heterocycles to complex biolipids, fatty acids, and many others. We are also highly skilled in all aspects of assay and antibody development, protein expression, crystallization, and structure determination.
Over the past thirty years, Cayman developed a deep knowledge base in lipid biochemistry, including research involving the arachidonic acid cascade, inositol phosphates, and cannabinoids. This knowledge enabled the production of reagents of exceptional quality for cancer, oxidative injury, epigenetics, neuroscience, inflammation, metabolism, and many additional lines of research.
Our organic and analytical chemists specialize in the rapid development of manufacturing processes and analytical methods to carry out clinical and commercial GMP-API production. Pre-clinical drug discovery efforts are currently underway in the areas of bone restoration and repair, muscular dystrophy, oncology, and inflammation. A separate group of Ph.D.-level scientists are dedicated to offering Hit-to-Lead Discovery and Profiling Services for epigenetic targets. Our knowledgeable chemists can be contracted to perform complete sample analysis for analytes measured by the majority of our assays. We also offer a wide range of analytical services using LC-MS/MS, HPLC, GC, and many other techniques.
Accreditations
ISO/IEC 17025:2005
ISO Guide 34:2009
Cayman is a leader in the field of emerging drugs of abuse, providing high-purity Schedule I-V Controlled Substances to federally-licensed laboratories and qualified academic research institutions for forensic analyses. We are certified by ACLASS Accreditation Services with dual accreditation to ISO/IEC 17025:2005 and ISO Guide 34:2009.