24(S),25-
24(S),25-
24(S),25-epoxy Cholesterol is an oxysterol and the most abundant oxysterol in mouse ventral midbrain.{43943} It activates liver X receptors (LXRs)...
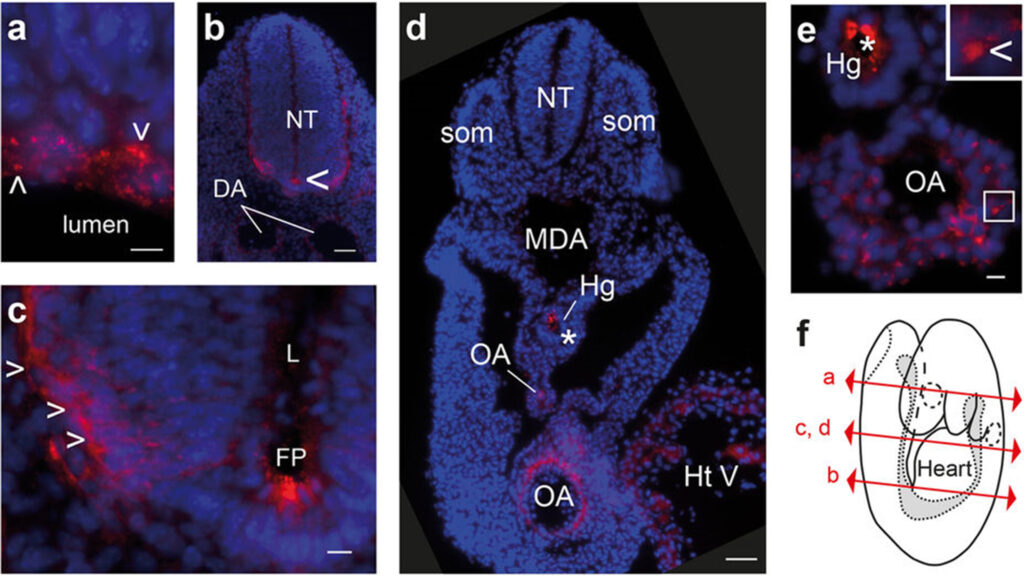
Prion Protein (PrP) and namely its abnormal isoform, partially resistant to proteinase K (PrPres), is the only specific molecular marker of the Transmissible Spongiform Encephalopathies (TSEs) such as Bovine Spongiform Encephalopathie (BSE) or its human form, the New Variant of Creutzfeld-Jakob disease.|This antibody recognises the human protein sequence within amino acids 145-152 (human numbering).
Territorial Availability: Available worldwide directly through Bertin or your local distributor
| Size | 200 µg |
|---|---|
| Shipping | dry ice |
| Host | Mouse |
| Antigen | This anti-prion protein (PrP) monoclonal antibody was raised against proteinase K treated and non-denatured scrapie-associated fibrils from Syrian hamster infected brain (263K). |
| Clone | Sha 31 |
| Isotype | IgG1k |
| Application(s) | 1 µg/mL). For western blot analysis of PrPc, dilute the antibody to a final concentration of 1 µg/mL., For EIA, Reconstitute the content of the vial in 1 mL of water., the optimal working dilution must be determined empirically (currently be-tween 0.1 |
| Specificity | PrPc (+), PrPsc (nda) Hamster, (+) Mouse, (nda) Bovine, (+) Ovine, (+) Human. |
| Application media | |
| Formulation | lyophilized IgG with BSA |
| Custom code | 3822000000 |
| UNSPSC code | 12352203 |
24(S),25-epoxy Cholesterol is an oxysterol and the most abundant oxysterol in mouse ventral midbrain.{43943} It activates liver X receptors (LXRs)...
Cytosolic PGE synthase (cPGES) is a glutathione-dependent enzyme with a predicted size of 18.6 kDa (23 kDa on SDS-PAGE). The...
Proprotein convertase subtilisin kexin 9 (PCSK9) is a member of the subtilisin serine protease family with an important role in...
This mixture contains primary prostaglandins produced from arachidonic acid and dihomo-?-linolenic acid. Contents: Prostaglandin E1, Prostaglandin E2, Prostaglandin F1?, 6-keto...
GST-tag Polyclonal Antibody (FITC) is a probe for the immunochemical detection of GST tags on recombinant proteins. Recombinant proteins are...
Protein phosphorylation is an important post-translational modification that serves many key functions to regulate a protein’s activity, localization, and protein-protein...
CD34 is a transmembrane phosphoglycoprotein and sialomucin protein that is commonly used as a marker for hematopoietic progenitor cells.{59706,59704} It...
(+)-D-threo-PDMP is a ceramide analog and is one of the four possible stereoisomers of PDMP (Item No. 62595).{11392} (+)-D-threo-PDMP is...
GPR17 is a G protein-coupled receptor that has been identified as a dualistic receptor recognizing signals from two unrelated chemical...
The Cayman COX Inhibitor Pack contains a combination of frequently used cyclooxygenase (COX) inhibitors. Each kit contains aspirin, the archetype...
This mixture contains the primary COX products produced by most mammalian tissues. Contents: Prostaglandin D2, Prostaglandin E2, 6-keto Prostaglandin F1?,...
This rabbit anti-mouse IgA FITC is used as the ‘secondary antibody’ for immunostaining experiments where the primary antibody is mouse...
Adenosine Receptor A2A is a multi-pass membrane protein that is normally localized to the plasma membrane.{15465} This receptor is part...
This mixture contains the primary metabolites of prostaglandins (PGs) D2, E2, and F2?. Contents: 13,14-dihydro-15-keto PGD2, 13,14-dihydro-15-keto PGE2, 11?-PGF2?, 13,14-dihydro-15-keto...
The cyclopentenone prostaglandin HPLC mixture contains all of the major UV-absorbing cyclopentenone prostaglandins and their precursors supplied in methyl acetate....
Secretory phospholipase A2 (sPLA2) (Type IIA) is a calcium-dependent PLA2 superfamily member that is encoded by PLA2G2A in humans.{56210} It...
This mixture contains the characteristic metabolites of both PGI2 and TXA2. Contents: Thromboxane B2, 11-dehydro Thromboxane B2, 6-keto Prostaglandin F1?,...
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a basic leucine zipper transcription factor encoded by NFE2L2 in humans that...