GST-tag Polyclonal Antibody (FITC)
GST-tag Polyclonal Antibody (FITC)
GST-tag Polyclonal Antibody (FITC) is a probe for the immunochemical detection of GST tags on recombinant proteins. Recombinant proteins are...
€536.00
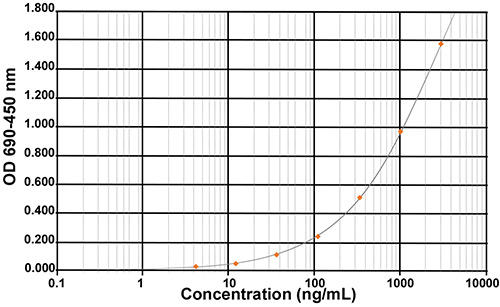
Enzyme ImmunoAssay (EIA) is a technique to detect and quantify antigens (proteins, hormones…) or antibodies in samples. It relies on the ability of an antibody to bind a specific antigen. Either the antibody or the antigen is labelled with an enzyme whose substrate is a chromogen or a fluorogen converted in a measurable product (color or fluorescence).|Enzyme-linked Immunosorbent Assay (ELISA) is a type of EIA using a solid phase (ex: microtiter plate) coated with an antigen immobilizing the molecule to detect. Over the time, scientists have extended the term ELISA to EIAs using an antibody coating the solid phase. That explains why our EIA kits using coated antibodies are also called ELISA kits.|Impurity assessment is a key step during the drug development production of recombinant proteins, including therapeutic proteins. Specific impurities coming from the cells mediating the protein expression, known as Host Cell Proteins (HCP) are generated and need to be removed. This kit is intended for use in assessing relative quantities of E. coli HCP in manufactured or research bioproducts.|Polyclonal antibodies used in this kit have been generated against several strains of E.coli and specifically selected for their recognition of a large spectrum of E. coli proteins. Thus, this kit can be considered as generic and allows a relative-quantitative determination of E. coli HCP in many types of samples, such as samples issued from the purification process (HCP clearance), process control, quality control, or product release.|Using this kit, HCP concentration is measured in ng/ml (HCP equivalent is extrapolated from a standard curve). Conventionally, the HCP content in a product will finally be expressed in ng/mg, where ng represents HCP mass and mg represents the product mass. Note that, contrary to the concentration measurement of the product, the HCP signal is only reflective of antibody binding and does not strictly reflect the mass of HCP.|This kit has been successfully validated for recovery and precision using reconstituted HCP samples and tested against different final products. Given the diversity of final products, all potential matrix effects cannot be known and it is recommended that you test the suitability of the kit for use with your own HCP samples in your laboratory.|This kit should be used as one part of your complete HCP analysis.
Territorial Availability: Available worldwide directly through Bertin or your local distributor
Technical Warning: Check the Additional Items Required section of this kit booklet to verify if UltraPure Water (Milli-Q or equivalent) is needed for this assay
| Size | 96 wells |
|---|---|
| Shipping | wet ice |
| Sample volume | 100 µL |
| Tracer | HRP |
| Detection limit | |
| Standard Curve Range | 4,1 – 3000 ng/mL |
| Custom code | 3822000000 |
| UNSPSC code | 41116104 |
GST-tag Polyclonal Antibody (FITC) is a probe for the immunochemical detection of GST tags on recombinant proteins. Recombinant proteins are...
TAF10 is one of many protein factors or coactivators associated with RNA polymerase II activity.{16903} One vial of this peptide...
To be used in conjunction with Cayman’s FABP3 polyclonal antibody (Catalog No. 10233) to block protein-antibody complex formation during immunochemical...
This mixture contains the primary COX products produced by most mammalian tissues. Contents: Prostaglandin D2, Prostaglandin E2, 6-keto Prostaglandin F1?,...
To be used in conjunction with Cayman’s NAPE-PLD polyclonal antibody (aa 6-20) (Item No. 10306) to block protein-antibody complex formation...
Histone H2A is a core histone that forms a dimer with histone H2B.{46631} Two histone H2A/H2B dimers form an octameric...
The Cayman COX Inhibitor Pack contains a combination of frequently used cyclooxygenase (COX) inhibitors. Each kit contains aspirin, the archetype...
Prostaglandin I synthase (PGIS) catalyzes the isomerization of PGH2 to PGI2. PGI2 (prostacyclin) is a potent vasodilator and inhibitor of...
Protein phosphorylation is an important post-translational modification that serves many key functions to regulate a protein’s activity, localization, and protein-protein...
This mixture contains the characteristic metabolites of both PGI2 and TXA2. Contents: Thromboxane B2, 11-dehydro Thromboxane B2, 6-keto Prostaglandin F1?,...
Programmed cell death protein 4 (PDCD4) levels are elevated during apoptosis and absent in many cancer samples.{16340,16167} Loss of PDCD4...
Proprotein convertase subtilisin kexin 9 (PCSK9) is a member of the subtilisin serine protease family with an important role in...
The cyclopentenone prostaglandin HPLC mixture contains all of the major UV-absorbing cyclopentenone prostaglandins and their precursors supplied in methyl acetate....
This mixture contains primary prostaglandins produced from arachidonic acid and dihomo-?-linolenic acid. Contents: Prostaglandin E1, Prostaglandin E2, Prostaglandin F1?, 6-keto...
Sirtuin 5 (SIRT5) is an enzyme that catalyzes the NAD-dependent removal of malonyl, succinyl, and glutaryl groups from target proteins.{55107,55108}...
Inositol hexakisphosphate kinase 2 (IP6K2) is a cytoplasmic kinase that catalyzes the conversion of IP6 to diphosphoinositol pentakisphosphate in the...
This mixture contains the primary metabolites of prostaglandins (PGs) D2, E2, and F2?. Contents: 13,14-dihydro-15-keto PGD2, 13,14-dihydro-15-keto PGE2, 11?-PGF2?, 13,14-dihydro-15-keto...
Platelet-activating factor (PAF) is an important lipid mediator involved in inflammation. PAF-acetylhydrolase (PAF-AH) is an extracellular phospholipase A2 which hydrolyzes...